RiVive: Uses, Benefits, Dosage, Side Effects Interactions
What is RiVive?
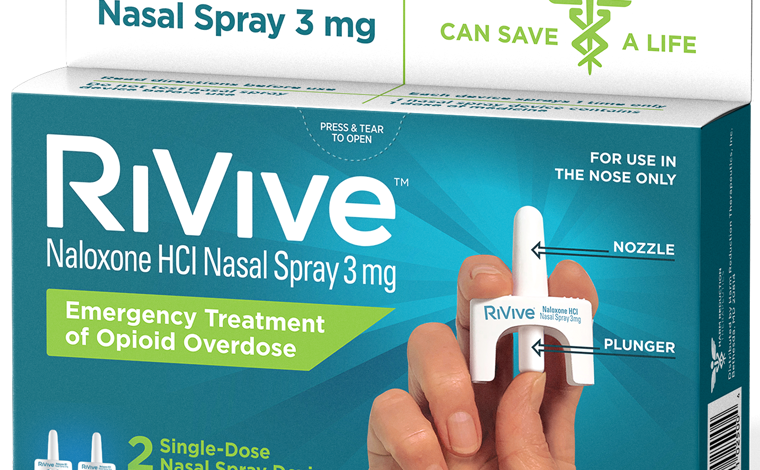
RiVive is a nasal spray medication containing 3 mg of Naloxone, designed for the emergency treatment of opioid overdose to prevent fatalities. Unlike some other naloxone products that require a prescription, RiVive will be available over-the-counter (OTC), making it more accessible to the general public. The company responsible for RiVive is Harm Reduction Therapeutics (HRT), Inc., a nonprofit pharmaceutical company, which aims to provide the medication at little to no cost to ensure widespread availability.
As an opioid antagonist, RiVive works rapidly to counteract the effects of opioids. It competes for opiate receptors in the brain and other parts of the body, effectively reversing the dangerous effects of opioids. By doing so, RiVive helps improve the patient’s breathing, alertness and normalizes their blood pressure.
In cases where opioid overdose is suspected and signs of slowed breathing, severe sleepiness, or loss of consciousness are evident, RiVive should be administered as soon as possible. However, it is crucial to remember that after administering the nasal spray, the individual must still receive immediate medical attention, even if they regain consciousness.
RiVive is effective against a range of opioids, including codeine, oxycodone (Oxycontin), heroin, morphine, hydrocodone (Vicodin, Lortab), fentanyl, methadone, oxymorphone, meperidine, tramadol, buprenorphine, and hydromorphone.
The expected release date for RiVive nasal spray is early 2024, following its FDA approval on July 28, 2023. This medication represents a significant advancement in addressing the opioid crisis and has the potential to save countless lives by providing timely and readily available overdose-reversal treatment to those in need.
RiVive Benefits
RiVive nasal spray offers several significant benefits in addressing opioid overdose emergencies and combating the opioid crisis:
1. Rapid Reversal of Opioid Overdose: RiVive is a powerful opioid antagonist that works quickly to reverse the effects of opioids. When administered promptly during an opioid overdose, it can restore normal breathing and consciousness, potentially saving the person’s life.
2. Increased Accessibility: Being an OTC medication, RiVive is more readily available to the general public without the need for a prescription. This increased accessibility empowers individuals, caregivers, and bystanders to intervene in overdose situations promptly.
3. Potential for Wide Distribution: RiVive is expected to be available at low cost or even free of charge since Harm Reduction Therapeutics (HRT), Inc., the manufacturer, is a nonprofit pharmaceutical company. This affordability enhances its accessibility to vulnerable populations, including those with limited resources or at higher risk of opioid overdose.
4. Non-Addictive: Naloxone, the active ingredient in RiVive, is non-addictive and has no potential for abuse. It solely functions to reverse opioid effects temporarily and does not produce any euphoria or habit-forming properties.
5. Easy to Administer: RiVive is administered through a nasal spray, which makes it easy to use, even for individuals without medical training. The simple administration process allows for quick intervention during emergencies.
6. Potential to Save Lives: By effectively reversing opioid overdoses, RiVive can save lives and prevent fatalities associated with opioid misuse or accidental overdose.
7. Reducing Stigma: The availability of OTC naloxone like RiVive helps reduce the stigma associated with opioid overdose and substance use disorders. It normalizes the concept of having naloxone on hand, similar to keeping a first-aid kit for other emergencies.
8. Public Health Impact: RiVive’s approval and availability align with the FDA’s efforts to prioritize access to naloxone products, providing a critical tool in protecting public health and combating the opioid crisis.
How to use RiVive
Using RiVive properly is crucial to ensure effective treatment during an opioid overdose emergency. Follow these steps for the correct administration of RiVive:
1. Read Instructions: Carefully read all instructions on the label, medication guides, or instruction sheets that come with RiVive. Familiarize yourself with the proper usage and guidelines before an emergency occurs.
2. Seek Professional Advice: If you are a caregiver or family member responsible for administering RiVive, consult your doctor, pharmacist, or other healthcare provider if you have any questions or need further clarification.
3. One-Time Use: Each RiVive nasal spray device contains a single dose of the medication and cannot be reused. Do not attempt to test the spray before use.
4. Positioning: Hold the nasal spray device with your thumb on the plunger and a finger on each side of the nozzle.
5. Administration: Insert the nozzle of the nasal spray gently into one nostril of the person experiencing the opioid overdose. Press the plunger firmly to administer the medication.
6. Emergency Assistance: Following the administration of RiVive, immediately seek emergency medical help by calling 911.
7. Wait Time: After giving the first dose of RiVive, wait for 2 to 3 minutes to allow the medication to take effect. If the person regains consciousness but becomes drowsy again, you can administer an additional dose.
8. Stay with the Person: Stay with the individual who experienced the overdose until medical assistance arrives, even if they regain consciousness.
9. Second Dose: If the person does not wake up 2 to 3 minutes after the first dose, administer a second dose using a new RiVive nasal spray device. Subsequent doses can be given every 2 to 3 minutes in alternating nostrils until emergency medical assistance arrives.
10. Continued CPR: Do not assume that the overdose episode has ended if symptoms improve. If necessary, perform CPR (cardiopulmonary resuscitation) while waiting for emergency help to arrive.
Dosage Information
• Usual RiVive nasal spray dose for opioid overdose: Administer 1 RiVive spray intranasally into 1 nostril.
• If the desired response is not achieved after 2 or 3 minutes, give a second dose using a new RiVive nasal spray device.
• Additional doses may be administered every 2 to 3 minutes in alternating nostrils until emergency medical assistance arrives.
Remember, it is essential to act swiftly and responsibly during an opioid overdose emergency. RiVive provides a crucial tool in saving lives, but prompt emergency medical assistance is still necessary for proper care and follow-up treatment.
Can a pregnant or breastfeeding woman use RiVive?
Pregnant and breastfeeding women should exercise caution when considering the use of RiVive. It is essential to inform your healthcare provider if you are pregnant or breastfeeding before using this medication.
Using RiVive during pregnancy may cause opioid withdrawal effects in the unborn baby. However, it is crucial to prioritize the treatment of an opioid overdose in the mother, as an untreated overdose can be life-threatening to both the mother and the baby. In the event of an overdose, emergency medical assistance must be sought immediately, and all medical caregivers should be informed of the pregnancy.
If a pregnant woman uses opioid medicine during pregnancy, the baby may become dependent on the drug. This can lead to life-threatening withdrawal symptoms in the baby after birth. Babies born dependent on opioids may require medical treatment for several weeks.
During emergencies, a woman may not be able to communicate her pregnancy or breastfeeding status to caregivers. Therefore, it is vital to ensure that any healthcare professional caring for the pregnancy or the baby is made aware if RiVive was administered.
What should I avoid while taking RiVive?
While taking RiVive, it is essential to be aware of certain precautions to ensure the safety and effectiveness of the medication. Here are some things to avoid:
1. Leaving a Person Alone: After administering RiVive to someone experiencing an opioid overdose, avoid leaving them alone. Naloxone can reverse the effects of opioids, which may cause the person to become alert and conscious suddenly. They might be disoriented or confused, and their thinking or reactions could be impaired. Staying with the person and providing reassurance until emergency medical help arrives is crucial.
2. Mixing with Other Drugs: Avoid using RiVive in combination with other medications or substances without proper medical guidance. Naloxone can interact with certain medications, and the effects may vary. Always inform your healthcare provider about any other medications you are taking to avoid potential drug interactions.
3. Self-Medication: Do not use RiVive for purposes other than treating opioid overdose. It is specifically designed for emergency use in opioid emergencies. Using RiVive in non-emergency situations or without proper indications can be harmful.
4. Storing Properly: Ensure that RiVive nasal spray is stored correctly according to the instructions provided. Keep it in a safe place, out of reach of children and unauthorized individuals.
5. Handling with Care: Handle the RiVive nasal spray device with care and avoid dropping or damaging it. Proper handling ensures the medication is effective when needed.
RiVive Side Effects
Common side effects of RiVive may include:
• Nausea
• Vomiting
• Sweating
• Increased heart rate
• Blood pressure changes
• Agitation
• Tremors
• Withdrawal symptoms in opioid-dependent individuals
It is essential to note that these side effects are generally short-lived and occur due to the sudden reversal of opioid effects. The benefits of using RiVive to save a life during an opioid overdose far outweigh the potential side effects.
Always follow the instructions provided by your healthcare provider and the medication’s packaging for the safe and appropriate use of RiVive. If you experience any unexpected or severe side effects, seek immediate medical attention.
What other drugs can interact with or affect RiVive?
RiVive can interact with other medications and substances. Some interactions can reduce the effectiveness of naloxone or may lead to adverse effects. It’s essential to be cautious and inform healthcare providers about all the medications, supplements, and recreational drugs you are taking to ensure the safe and effective use of naloxone. Here are some common drug interactions with naloxone:
1. Opioids: Naloxone is an opioid antagonist, meaning it blocks the effects of opioids. When used to reverse opioid overdose, it can cause sudden withdrawal symptoms in individuals who are dependent on opioids. Therefore, it is essential to be aware of the patient’s opioid use and provide appropriate medical care following naloxone administration.
2. Opioid Analgesics: Naloxone can reduce the effects of opioid analgesics when used simultaneously. This may be of concern if naloxone is inadvertently given to manage pain in someone using opioid pain medication.
3. Partial Agonist Opioids: Drugs like buprenorphine, which have partial agonist activity at opioid receptors, can have reduced effects when naloxone is administered concurrently.
4. Mixed Agonist/Antagonist Opioids: Naloxone can precipitate withdrawal symptoms in individuals taking mixed agonist/antagonist opioids, such as pentazocine or nalbuphine.
5. Nalmefene: Nalmefene is another opioid antagonist used to reverse opioid overdose. Concurrent use of naloxone and nalmefene is not recommended, as they may counteract each other’s effects.
6. Other Medications: Some drugs may interact with naloxone in ways that have not been extensively studied or documented. Always inform your healthcare provider about all the medications you are taking, including prescription, over-the-counter, and herbal products.
7. Recreational Drugs: Certain recreational drugs, such as illicit opioids or other substances acting on opioid receptors, can interact with naloxone. This interaction may affect naloxone’s ability to reverse opioid overdose or cause unpredictable reactions.
Always seek guidance from healthcare professionals, such as doctors or pharmacists, to ensure the safe use of naloxone in conjunction with other medications. Providing accurate information about your medical history and drug use is vital to avoid potential complications and to ensure naloxone’s effectiveness during opioid overdose emergencies.