FDA Expands Use of AstraZeneca’s Farxiga to Broader Range Heart Failure
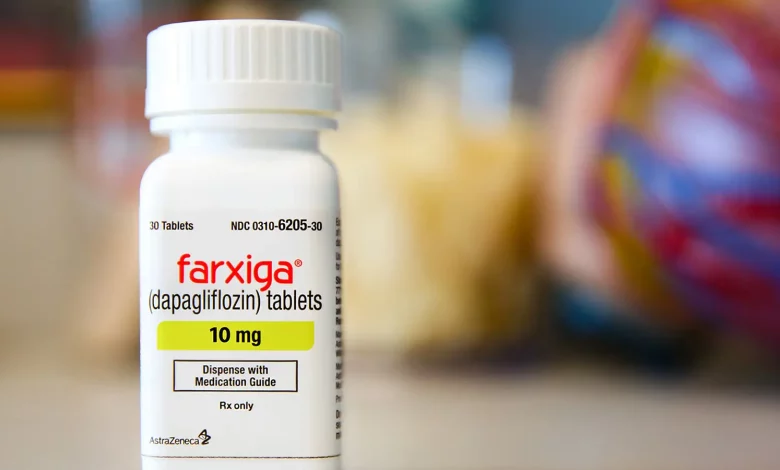
Dapagliflozin (Farxiga, AstraZeneca), a sodium-glucose cotransporter 2 (SGLT2) inhibitor, has received an expanded indication from the US Food and Drug Administration (FDA) for the treatment of heart failure (HF) across the entire range of left-ventricular ejection fraction (LVEF). This includes HF with preserved ejection fraction (HFpEF) and mildly reduced ejection fraction (HFmrEF), in addition to heart failure with reduced ejection fraction (HFrEF), for which the drug was previously approved in the US.
The decision to expand the indication was based on data from the DELIVER trial, a phase 3 study involving over 6200 patients, which showed that dapagliflozin provided significant clinical benefits for patients with HF regardless of their left-ventricular function. The primary composite endpoint of cardiovascular (CV) death or worsening HF was significantly reduced by dapagliflozin treatment for patients with HFmrEF or HFpEF, as reported earlier by theheart.org | Medscape Cardiology.
Furthermore, a pooled analysis of the DAPA-HF and DELIVER phase 3 trials indicated that dapagliflozin consistently reduced the combined endpoint of CV death or HF hospitalization across the full range of LVEF. The European Commission had already expanded the indication for dapagliflozin (Forxiga) to include HF across the full spectrum of LVEF in February, according to theheart.org | Medscape Cardiology.
Dapagliflozin was initially approved in 2014 to improve glycemic control for patients with diabetes mellitus and has also been approved for use by patients with chronic kidney disease.