FDA Adds Boxed Warning for Prolia in Advanced CKD Due to Severe Risk Of Hypocalcemia
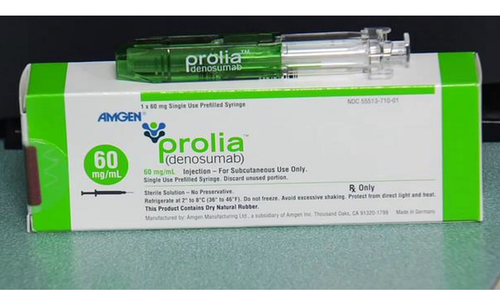
The U.S. Food and Drug Administration (FDA) has issued a Drug Safety Communication regarding Prolia (denosumab), an osteoporosis medication. Following a comprehensive review, the FDA has determined that Prolia increases the risk of severe hypocalcemia, characterized by very low blood calcium levels, particularly in patients with advanced chronic kidney disease (CKD), especially those on dialysis. Severe hypocalcemia is found to be more prevalent in patients with CKD who also suffer from mineral and bone disorder (CKD-MBD). Instances of severe hypocalcemia in advanced CKD patients taking Prolia have led to serious harm, including hospitalization, life-threatening events, and death.
As a response to these findings, the FDA is implementing a Boxed Warning, its most prominent warning, to the Prolia prescribing information. This warning will communicate the heightened risk of severe hypocalcemia in patients with advanced CKD. The updated information includes recommendations for reducing this risk, such as careful patient selection for Prolia treatment, increased monitoring of blood calcium levels, and other strategies. This information will be integrated into the patient Medication Guide and the Prolia Risk Evaluation and Mitigation Strategy (REMS), a drug safety program mandated by the FDA.
Background:
Prolia, a monoclonal antibody initially developed for postmenopausal osteoporosis treatment, has received subsequent approvals for various conditions affecting bone health. It operates by inhibiting a protein called RANK (receptor activator of nuclear factor kappa beta) to prevent the breakdown of bone cells known as osteoclasts.
Common side effects of Prolia include back pain, muscle pain, and pain in the arms or legs. Administered as a subcutaneous injection every six months by a healthcare professional, Prolia may lower blood calcium levels.
Recommendations:
For Patients:
1. Consult with Healthcare Professionals: Patients considering Prolia for osteoporosis treatment should discuss their kidney function and the risk of severe hypocalcemia with their healthcare professional.
2. Maintenance of Calcium and Vitamin D Intake: Patients currently taking Prolia should ensure adequate calcium and vitamin D intake while receiving the medication.
3. Monitoring for Advanced Kidney Disease Patients: Advanced kidney disease patients, especially those on dialysis treated with Prolia, should undergo frequent monitoring of blood calcium levels, particularly in the initial weeks after each injection.
4. Communication with Healthcare Professionals: Patients should not discontinue Prolia without consulting their healthcare professional, as stopping the treatment may increase the risk of bone fractures.
For Healthcare Professionals:
1. Assessment of Kidney Function: Before prescribing Prolia, healthcare professionals should assess patients’ kidney function, particularly in those with advanced CKD.
2. Consideration of Severe Hypocalcemia Risk: For patients with advanced CKD, especially those on dialysis, healthcare professionals should weigh the risk of severe hypocalcemia with Prolia against other available osteoporosis treatments.
3. Expertise in CKD-MBD Management: Treatment with Prolia in advanced CKD patients, including those on dialysis, should involve healthcare providers with expertise in the diagnosis and management of CKD-MBD.
4. Monitoring and Management Post-Prolia Administration: After Prolia administration, close monitoring of blood calcium levels and prompt management of hypocalcemia is crucial to prevent complications.
Patients and healthcare professionals are urged to promptly report any symptoms consistent with hypocalcemia, such as confusion, seizures, irregular heart rhythm, fainting, face twitching, uncontrolled muscle spasms, weakness, tingling, or numbness in parts of the body.
The FDA emphasizes that these measures are essential for the safe use of Prolia in patients with advanced CKD, and compliance with these recommendations is crucial to minimize the risk of severe hypocalcemia and associated complications.