Soterios Pharma Wraps Up Phase II Enrollment for STS-01 in Alopecia Areata
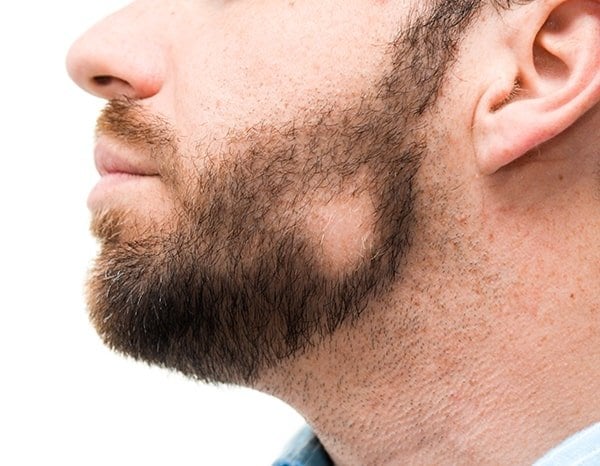
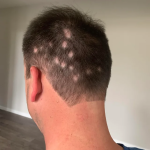
Soterios Pharma, a clinical-stage pharmaceutical company, has announced the completion of target enrollment for its Phase II trial with STS-01, a treatment for mild to moderate (patchy) alopecia areata. The final results of this study are expected to be available in the second quarter of 2024.
David Fleet, CEO of Soterios Pharma, highlighted the significance of this milestone, stating, “This represents an important milestone in the development of STS-01 as the first approved treatment for patients with mild-to-moderate alopecia areata. While there are now options available for severe patients, there are still no approved treatments for patients with patchy disease – and yet they represent over half of the alopecia areata population, and the quality-of-life impact can be devastating. We are developing STS-01 as an effective and convenient topical treatment to address this unmet need.”
The Phase II trial is a randomized, placebo-controlled study that is assessing the safety and efficacy of STS-01 when applied over a 6-month period. A total of 158 participants have been randomly assigned to receive one of four doses of STS-01 or a placebo, with the effectiveness of the treatment being evaluated based on improvements in the SALT (Severity of Alopecia Tool) score.
About Soterios Pharma: Soterios Pharma is a UK-based clinical-stage life sciences company with a focus on developing and commercializing medicines for dermatological diseases that have significant unmet needs. The company’s approach is based on deep scientific knowledge of medical dermatology to identify opportunities to improve patients’ lives.
About STS-01: STS-01 is a product that utilizes a mechanism with a well-established safety profile in dermatology. It works by modulating the inflammatory response and the proliferation of T-cells through disruption of the signaling pathways. There is extensive pre-clinical and clinical evidence to support its efficacy and safety profile in the treatment of alopecia areata. STS-01 is being developed as a topical treatment for mild to moderate alopecia areata, with a SALT score of less than 50. This population represents over half of the 700,000 alopecia areata patients in the United States.