The Controversy Surrounding Fish Antibiotics: Are They Safe for Human Use?
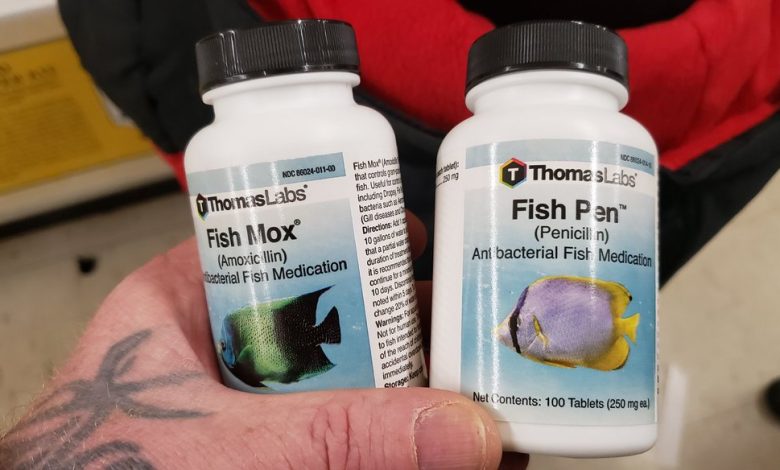
In recent years, there has been a growing trend among some individuals to turn to fish antibiotics as an alternative source for obtaining medications. With concerns about the accessibility and affordability of healthcare, some people are exploring unconventional options.
Fish antibiotics, originally formulated for aquatic animals, have become a subject of controversy, raising questions about their safety and efficacy for human use. In this article, we will delve into the reasons behind this trend, examine the risks and potential benefits, and explore the broader implications for public health.
The Rise of Fish Antibiotics
The surge in interest in fish antibiotics among certain segments of the population can be attributed to various factors. One of the primary drivers is the ease of availability. Fish antibiotics are readily accessible at pet stores, online retailers, and aquarium supply shops without the need for a prescription. This convenience has led some individuals to self-prescribe and self-medicate with these products, often without consulting healthcare professionals.
A recent study in PlosOne (Zhang et al. 2020) looked at the availability of OTC antibiotics on internet sites from the US, and tried to infer information about purchase for self-medication. They did that by looking at comments and “likes” that the comments received on the websites.
- Not surprisingly, a variety of medically-important antimicrobials were available for fish: amoxicillin cephalexin, metronidazole, ciprofloxacin, penicillin, clindamycin, doxycycline, erythromycin and trimethoprim-sulfa.
- Overall 2.4% of comments and reviews for drugs on these sites were potentially associated with human use. Comments about human use received a lot more “likes” – 9.2 per review versus 0.5 per review not related to suspected human use.
- The antibiotics that had the most human use-related reviews were erythromycin (8.3%), clindamycin (5%), ciprofloxacin (3.4%), penicillin (3.3%) and doxycycline (2.8%).
- The greatest number of “likes” was for use of ciprofloxacin (ugh…).
The Antibiotic Resistance Crisis
Before delving into the safety concerns associated with fish antibiotics, it is crucial to understand the broader context of antibiotic use. The global rise of antibiotic resistance poses a significant threat to public health. Misuse and overuse of antibiotics contribute to this crisis by fostering the development of resistant strains of bacteria. In this context, the uninformed use of fish antibiotics by humans raises serious concerns about the potential exacerbation of antibiotic resistance.
Quality and Regulation
One of the main challenges in assessing the safety of fish antibiotics for human use lies in the lack of regulation and quality control. Fish antibiotics are not subject to the same rigorous standards and testing procedures as medications intended for human consumption. The formulations, manufacturing processes, and overall quality of these products may vary widely, leading to potential inconsistencies in their safety and efficacy.
Cross-Species Suitability
Fish antibiotics are specifically designed for aquatic animals, and their efficacy and safety profiles are based on the physiological characteristics of fish. The question arises: can antibiotics formulated for fish be safely and effectively extrapolated to humans? The differences in anatomy, metabolism, and susceptibility to certain infections between fish and humans make this a complex and largely unexplored area of concern.
Potential Risks of Fish Antibiotics for Humans
1. Inaccurate Dosage: Fish antibiotics are typically sold in formulations and dosages appropriate for fish. When used by humans, achieving accurate dosing becomes a challenge. Inappropriate dosages can lead to inadequate treatment, contributing to antibiotic resistance, or, conversely, increase the risk of adverse effects.
2. Unknown Allergens: The excipients and fillers used in fish antibiotics may differ from those used in human medications. Individuals allergic to specific components present in fish antibiotics may experience allergic reactions, ranging from mild skin rashes to severe anaphylaxis. A study on fish antibiotics reported that all products contained the equivalent of not less than 90.0% and not more than 120.0% of their labeled contents, within the USP standards. Although no major impurities were identified, there was evidence of several as-yet unidentified excipient ingredients
3. Contamination and Purity Issues: The manufacturing processes for fish antibiotics may not adhere to the same standards as those for human medications. Contamination issues, such as the presence of impurities or unintended substances, could pose serious health risks.
Balancing Act: Potential Benefits and Responsible Use
While the risks associated with using fish antibiotics for human purposes are evident, some argue that, when used responsibly and under the guidance of a healthcare professional, there may be instances where these medications can be a viable option. In remote or emergency situations where access to conventional medical care is limited, having antibiotics on hand could potentially save lives. However, it is essential to stress that responsible use includes consulting with a healthcare professional for proper diagnosis, dosage determination, and monitoring of treatment.
Conclusion
The use of fish antibiotics for human consumption is a complex and contentious issue that intertwines concerns about antibiotic resistance, regulation, and the potential risks of cross-species medication use.
Studies have shown that self-medication and antibiotic utilization without healthcare oversight may lead to delayed appropriate treatment, transmission of communicable infections, untoward adverse events, and contribute to antimicrobial resistance.
While some individuals may see fish antibiotics as a pragmatic solution to healthcare challenges, the overall lack of regulation, quality control, and scientific evidence regarding their safety for humans raises significant red flags. In the face of a growing antibiotic resistance crisis, it is crucial for individuals to prioritize seeking professional medical advice and relying on approved and regulated medications to ensure both personal health and the collective well-being of society.

