FDA Warns Against the Use of Probiotics in Preterm Infants
The U.S. Food and Drug Administration (FDA) is issuing a critical warning to healthcare providers regarding the use of products containing live bacteria or yeast, commonly referred to as probiotics, in preterm infants within hospital settings. This advisory is crucial due to the potential risk of invasive, and in some cases, life-threatening diseases that can result from the use of probiotics in this vulnerable patient population.
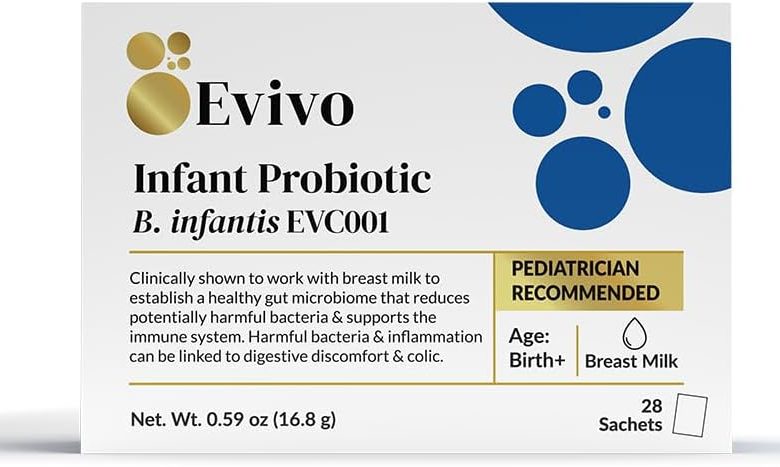
The FDA states that preterm infants administered probiotics are at an elevated risk of developing invasive diseases caused by the microorganisms contained in these products. As a tragic illustration of this risk, the FDA reports a devastating incident wherein a preterm infant with a birth weight of less than 1000 grams was administered the probiotic product Evivo with MCT Oil, developed by Infinant Health, as part of in-hospital care. Tragically, this infant subsequently developed sepsis caused by the bacterium Bifidobacterium longum, ultimately leading to the infant’s demise.
Specifics of the Probiotic Involved
Evivo with MCT Oil is a probiotic product deliberately formulated to contain the live bacterium Bifidobacterium longum subsp. infantis. Of utmost concern, genomic sequencing data conclusively demonstrate that the bacterium responsible for the sepsis in this infant was an exact genetic match to the strain contained in this specific probiotic.
Information on Probiotic Safety
The FDA provides further insight into the risks associated with probiotics in preterm infants. Notably, microorganisms present in probiotics have been reported in medical literature as potential culprits behind bacteremia or fungemia, sometimes resulting in severe clinical courses in very preterm or very low birthweight (VLBW) infants.
Moreover, the American Academy of Pediatrics has explicitly conveyed their reservations, stating that due to the lack of FDA-regulated pharmaceutical-grade products in the United States, conflicting data on safety and efficacy, and the potential for harm in a highly vulnerable population, current evidence does not support the routine, universal administration of probiotics to preterm infants, particularly those with a birth weight of less than 1000 grams.
Probiotic Products Not Approved as Drugs
The FDA stresses that no probiotic product has been approved by the agency for use as a drug or biological product in infants. It is acknowledged that some unapproved and unlicensed probiotics are nevertheless available for use in treating or preventing diseases or conditions in infants, including reducing the risk of necrotizing enterocolitis (NEC) in preterm infants. Healthcare providers must recognize that these products have not undergone the FDA’s rigorous premarket review evaluation for safety and effectiveness. Nor have they been subjected to the agency’s rigorous manufacturing and testing standards for drugs and biological products, including tests for extraneous organisms.
Requirements for Investigational New Drug Application (IND): Healthcare providers who administer probiotics containing live bacteria or yeast to treat, mitigate, cure, or prevent a disease or condition are reminded that they must submit an Investigational New Drug Application (IND) to the FDA.
Reporting Adverse Events: The FDA strongly encourages healthcare providers and consumers to report any adverse events following the use of probiotics. Reports should be submitted both to the product manufacturer using the contact information required to be on the product label and to the FDA directly. Reports can be filed online or by calling 1-800-FDA-1088.
For additional case reports and scholarly resources, please refer to the following references:
- Case reports of bacteremia and fungemia from probiotic use in preterm infants: Link, Link, Link, Link.
- Poindexter B. Use of probiotics in preterm infants. Pediatrics 2021;147(6):June 2021:e2021051485. Link
Source: FDA