Use of Unapproved Fat-Dissolving Injections Poses Serious Health Risks FDA Warns
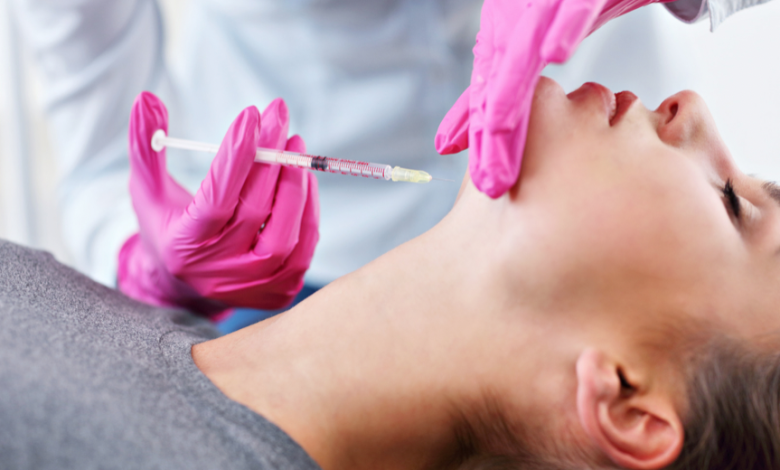
In a recent announcement, the Food and Drug Administration (FDA) has issued a stern warning about the potential dangers associated with the use of unapproved fat-dissolving injections, emphasizing the serious health risks posed by these products.
Individuals are cautioned against using such injections, as they have not been evaluated or approved by the FDA for safety and effectiveness.
How Fat-Dissolving Injections Work
Fat-dissolving injections, also known as injection lipolysis, involve the administration of a series of injections under the skin to break down fat cells in targeted areas. Despite the non-surgical nature of this procedure, it can lead to adverse reactions when performed with unapproved products.
Adverse Reactions from Unapproved Injections
Fat-dissolving injections not approved by the FDA are being sold online under various brand names, including Aqualyx, Lipodissolve, Lipo Lab, Kabelline, and others. The FDA has received reports of adverse reactions from consumers who have used these unapproved products, including:
• Permanent scars
• Serious infections
• Skin deformities
• Cysts
• Deep, painful knots
Specific Risks Associated with Unapproved Ingredients
The unapproved fat-dissolving injections often contain ingredients such as phosphatidylcholine (PPC) and sodium deoxycholate (DC), sometimes referred to as “PCDC injections.” These ingredients have not undergone FDA evaluation, posing a significant safety risk to consumers.
Risks from Improper Injection Techniques
Aside from the risks associated with unapproved ingredients, improper or unsafe injection practices by unlicensed personnel can increase the likelihood of scarring, skin infections, and serious complications. The correct administration of these injections depends on factors such as the number and location of injections, needle placement, and overall administration technique.
FDA-Approved Alternative
Kybella, a prescription drug containing deoxycholic acid, is the only FDA-approved fat-dissolving injectable drug. However, it is specifically approved for reducing fat under the chin in adults. Consumers are advised to consult a healthcare professional for FDA-approved injections and should refrain from purchasing fat-dissolving products online for self-administration.
Key Recommendations:
• Consult a healthcare professional for FDA-approved injections.
• Avoid purchasing fat-dissolving products online for self-injection.
• Report any adverse reactions promptly to your healthcare provider.
• Seek medical care if you are experiencing side effects from these injections.
Reporting Side Effects to FDA
Individuals are encouraged to report any adverse reactions or side effects from fat-dissolving injections, as well as other medications, to the FDA’s MedWatch program. Reports can be submitted online or by downloading the reporting form and faxing it to 1-800-FDA-0178.
The FDA emphasizes the importance of prioritizing patient safety and urges individuals to exercise caution when considering fat-dissolving injections, especially those obtained from unverified sources.
For more information, visit the FDA website or contact the FDA directly. SEE: How Long Do Kybella Side Effects Last?





