Johnson & Johnson Slashes Price of TB Drug in Low-Income Countries to Expand Access
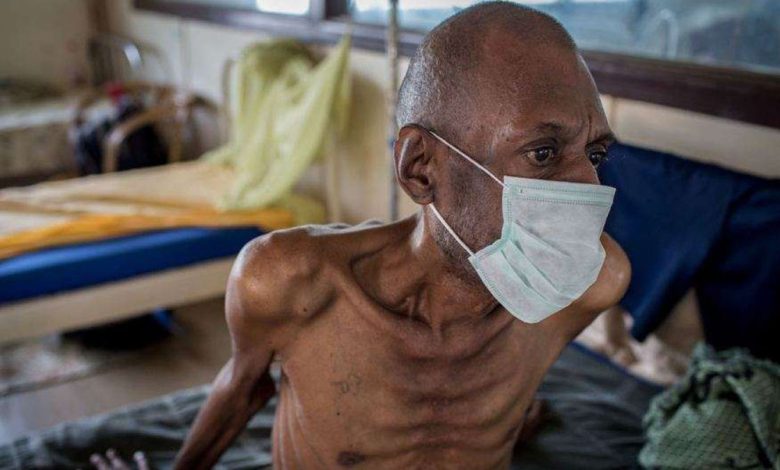
Seven weeks after announcing its decision to allow generic competition for the drug-resistant tuberculosis (TB) treatment Sirturo (bedaquiline) in low- and middle-income countries (LMICs), Johnson & Johnson (J&J) has taken a significant step by reducing the price of its own drug in those markets. Under the newly introduced pricing structure, J&J’s Sirturo will now cost $130 for a six-month course in LMICs, marking a substantial 55% reduction from its previous price. Concurrently, another pharmaceutical company, Lupin, has also lowered the cost of its generic version of bedaquiline by 33%, pricing it at $194. These price adjustments were unveiled by the United Nations-backed Stop TB Partnership.
This development follows J&J’s prior commitment to not enforce secondary patents on Sirturo in LMICs. This move facilitated the entry of generic competitors into various global markets, which was met with praise for potentially expanding access to life-saving TB treatment options.
The primary patent for Sirturo, a groundbreaking drug approved for drug-resistant TB treatment in 2012 after a four-decade gap, recently expired. However, J&J had secured additional patents for improvements to the drug’s formulation, effectively extending its patent protection until 2027. The decision to lower prices aims to enhance access to bedaquiline and aligns with global efforts to combat TB, with the goal of ending the disease by 2030, as stated by Atul Gawande, the assistant administrator of USAID’s Global Health Bureau.
The Stop TB Partnership estimates that approximately 450,000 individuals worldwide are affected by drug-resistant TB. The reduced prices for bedaquiline are projected to save $8 million over a 16-month period. This cost reduction will allow the organization’s Global Drug Facility to procure an additional 51,000 doses of the medication.
J&J’s resolution to forgo enforcing secondary patents emerged shortly after the company faced public criticism, largely ignited by a viral Twitter post from author John Green. The post garnered considerable attention, with 622,000 views within 24 hours. Green expressed concern that the pursuit of secondary patents, despite the potential to negatively impact hundreds of thousands of lives, could harm the company’s reputation. J&J responded by clarifying that it had already entered into an agreement with the Stop TB Partnership, effectively permitting generic competition.
Earlier in the year, Doctors Without Borders urged J&J to relinquish secondary patent protection in countries grappling with a high burden of TB. The organization posited that removing patent protection could lead to a two-thirds reduction in the price of bedaquiline. For nations like Nigeria, where drug-resistant TB is a significant health challenge, the reduced price for bedaquiline comes at a crucial juncture. According to Chukwuma Anyaike, the director of public health in Nigeria, the price reduction is particularly timely as the country aims to expand its treatment coverage and address the increasing demand for effective TB medications.





